I also have some work to do with the EC PPM's as those values are measured and not calculated. I can add as many products as I wish using the label #'s however with liquid ingredients I need to calculate the specific gravity requiring a sample to weigh. So I am working up a nutrient PPM calculator spreadsheet. While it doesn't give the complete picture it's a good starting point and better than following most other feeding tables that have you feeding too heavy for too long. I've not used one for over 5 year now.įor anybody who gets to grips with elemental ppm, the value tables in this link will be of good use as you can begin tailoring the varying ppm levels toward it. I can understand the use of ec meters in hydro as it can give you an idea of how heavy the plants are feeding, outside of a few uses like that I struggle to see the need. It's helpful to know how much N you have, how much P, etc. Working out elemental ppm gives accurate ppm levels and happier plants. So to add to your very helpful post, anybody who takes the time to work out the elemental ppm of the nutrients they are using (in the way the OP has done) will never have to use a ppm or ec meter again. Working things out as you have by actual weight is ''elemental ppm'' and is the most accurate way. Something I didn't know myself until recently is that ppm/ec meters have nothing to do with ''elemental ppm'' (kinda confusing right).

I didn't go into conversions of PPM from EC as thats really another subject and very simple. Please contribute if you see anything important I missed or any unfortunate errors in my work. I hope this helps explain PPM and I regret any errors in my math. 05 = 13.2 PPM of calciumīUT when we take into account the extra mass in the mL we multiply that by a specific gravity of 1.212 (according to my most likely not accurate measurement) and then we get 16 PPM of calcium by adding 1 mL of Micro to a gallon of water.ġ mL per gallon x 264.2 x. So if I do some math on the GH micro, 1mL per gallon, the label says it has 5% calcium.ġ mL per gallon x 264.2 x. Since the specific gravity of a nutrient isn't commonly listed on the label we must weigh a volume of the nutrient and calculate it's specific gravity.įor example, I just put 100mL of GH Micro in a graduated cylinder and it weighed about 121.2 grams. In this case you must take density into account. It gets more complicated when mixing by volume and not mass, liquid nutrients. Liquid Nutrients and Specific Gravity - Volume Measurements This means we have to take into account conversion factors to calculate the percent elemental P and K. Similarly, the labels lists the percentage K2O instead of the percent K. the fertilizer label lists the percentage P2O5 instead of the percent P. It should be noted that P & K are different animals depending on how they are put in the mix.īy convention in the U.S. Phosphorous and Potassium - Beasts of another nature Of Sulfur comes along with the 40 PPM of magnesium. Now that also brings some sulfur and we can calculate that, Epsom is 9.7% Magnesium and 13% Sulfur.Ĥ12 x 3.785 = 1560.8mg or 1.56 grams per gallon to get 40 PPM of Magnesium. So lets say you want 40 PPM of Magnesium from Epsom salt. To figure out how much of something to add to get a given PPM you divide the target PPM by the percentage The calcium nitrate also has 19% calcium.

So for basic dry (mass) calcs just take 264 times the percentage and you know how many PPM of something you get with a gram per gallon. Of nitrogen by adding 1 gram to a gallon. Grams per Gallon x 264.2007926023778 x Label Percentage = PPMĬalcium Nitrate is 15.5% Nitrogen therefore we get, Say we are adding half a gram of Calcium Nitrate to 1 Liter of H2O and we want to see how much calcium and nitrogen we added in PPM, we know the calcium nitrate is 19% Calcium.įirst get the mg per liter from gram per gallon: If we want to calculate how much of a given nutrient is in the mix using the label ingredient percentages we can make that happen with a little math. This is all fine and dandy for figuring out how much of everything is in your mix but what if you want to get more specific and determine how much of a given nutrient is present in your mix. Since different substances in the water will conduct electricity with varying degrees of resistance we must understand this number is only an estimate.
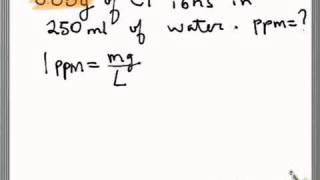
This is then converted to a PPM number with a conversion factor. PPM is commonly measured by a grower using an electronic pen which measures the electrical conductivity of the water (EC). Basically the idea is to quantify how much “stuff” is in your water. PPM (Parts Per Million) can seem like a mystery but it’s actually very simple. Understanding and Calculating Nutrient PPM
